
In a First: Has Science Cracked the Code on Chronic Pain?
Vita gazette – Research conducted in the USA has revealed that new findings regarding the source of pain in the brain can provide hope for patients suffering from chronic pain. In a First, UCSF Researchers Have Identified Chronic Pain States in Individuals Directly from Their Neural Activity.
Until now, there has been no objective measure of chronic pain and how severe it might be. Furthermore, because how chronic pain reorganises neural networks is unique to each individual, we need more personalised treatment strategies for dealing with it.
In a new study reported on May 22 in Nature Neuroscience, MD, PhD, associate professor of anaesthesia, neurology, and neurological surgery Prasad Shirvalkar and his colleagues looked for those biomarkers by implanting a neural recording device in the anterior cingulate cortex (ACC) and orbitofrontal cortex (OFC) of four people (three with post-stroke pain and one with phantom limb pain).
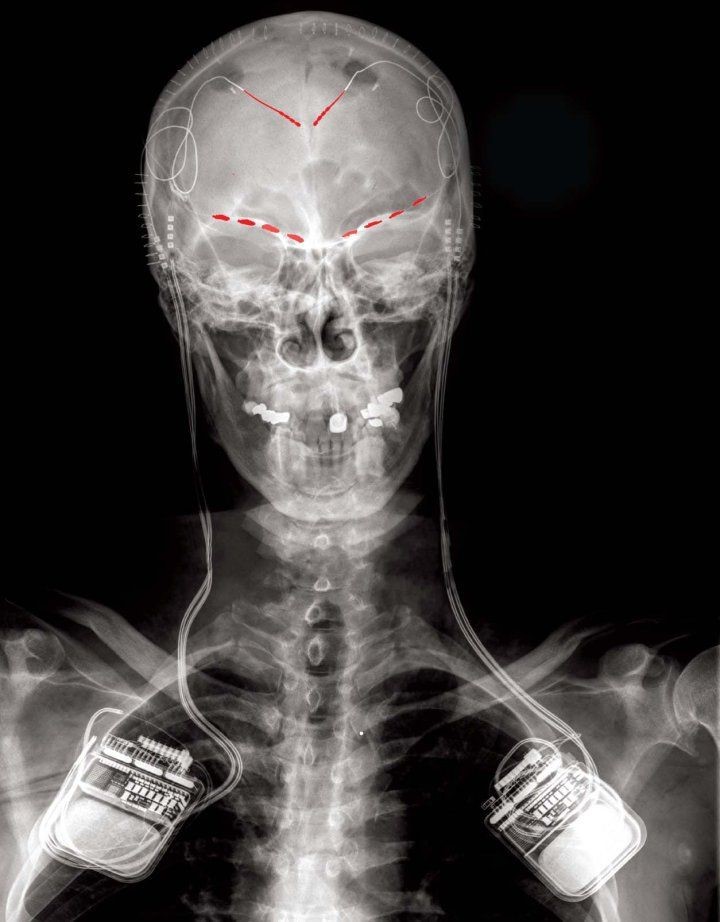
Frontal x-ray image of one study participant showing implanted brain recording electrodes (in red) connected to a bidirectional brain stimulating and recording implant on both sides of the body. Image by Prasad Shirvalkar
The study participants went about their daily lives for several months and reported pain scores about three times a day that described their pain severity and how it made them feel. Immediately after jotting down the reports, they used a remote control to record 30 seconds of neural activity from the device implanted in their brains.
Using machine learning models, the researchers were able to predict the reported pain levels with specific neural activity patterns in each patient, defining a unique neural signature for that person’s pain experience. They found that signals from the OFC were more strongly correlated with episodes of chronic pain than those from the ACC.
This is a significant milestone because it’s the first time that neural activity related to chronic pain has been measured in the real world over a clinically relevant period.
Your Brain on Different Kinds of Pain
In the lab, participants were exposed to a painful heat stimulus experiment to compare how chronic pain and acute pain are represented in the brain. This time, in two participants, the neural activity associated with pain came primarily from signals generated in the ACC. Unfortunately, it didn’t last as long as the sustained signs seen in the OFC during the recordings that correlated with chronic pain.
Despite its small sample size, this study provides the first direct evidence that acute and chronic pain have different neural representations within the same person’s brain. That evidence underlines what many doctors and patients know: treatments that help address acute pain, such as opioids, are less likely to be effective for chronic neuropathic pain.
Chronic pain in the brain
Animation showing activity in the brain coincides with phantom limb pain reported by a subject in the study. Participants coloured in a silhouette highlighting the location and degree of pain while researchers recorded the brain activity. The work provides the first direct evidence that acute and chronic pain have different representations in the brain.
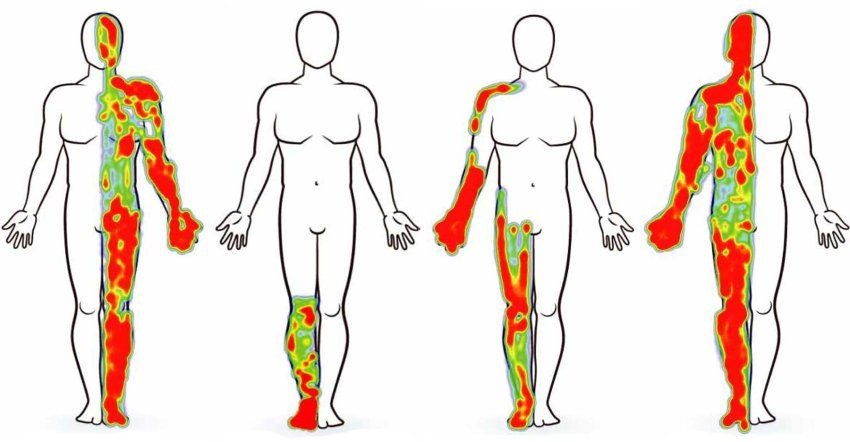
Personel maps of pain
During the study, patients drew their pain body diagrams. Below are charts from four different study participants showing the location and intensity of their pain. Green equals mild pain for the participant, while red equals severe pain. From left to right: post-stroke pain, phantom limb pain, post-stroke pain, post-stroke pain. Image by Prasad Shirvalkar.
Neural Networks Gone Awry
Developing personalised biomarkers of chronic pain has enormous implications for future therapies. For example, if researchers can define subtypes of chronic pain by their neural signatures, they can determine which signatures respond best to different treatments.
One possible treatment would be administering a therapeutic electrical current that disrupts the abnormal, long-lasting connections between neurons when acute pain transforms into chronic pain.
Chronic pain manifests very differently in different people, which makes it a perfect candidate for personalised neurostimulation.
Share:
